The Cecal Ligation and Puncture (C.L.P.) model is a widely used in vivo method for inducing polymicrobial sepsis in mice. By surgically ligating and puncturing the cecum, the model closely replicates key aspects of human sepsis and is commonly used to evaluate disease progression, immune responses, and treatment efficacy.
What Is C.L.P?
Cecal Ligation and Puncture (C.L.P.) is a controlled surgical procedure most commonly performed in male C57/Bl6 mice. The procedure targets the cecum, a pouch-like structure of the large intestine responsible for storing and breaking down undigested food material, allowing researchers to reliably induce a septic infection for a preclinical study. This model is widely used for antibacterial, cardiovascular and inflammatory targets.
Pre-Surgery Preparation (Day -1)
To prepare for the surgery, on Day -1 (the day before surgery), the mice are put under anesthesia, shaved, and receive an application of Nair around where the surgical site will be. This is done to minimize the risk of contamination from the mouse’s fur during the surgery. The area that is shaved is roughly 1.5” by 1.5” and is located along the left side of the mouse’s body, between the forelimb and hindlimb. After the Nair has been applied for 1-2 minutes, it is rinsed off, the area is wiped dry, and the mice are returned to their home cage to recover from anesthesia. They are now ready for surgery.
C.L.P. Surgery Procedure (Day 0)
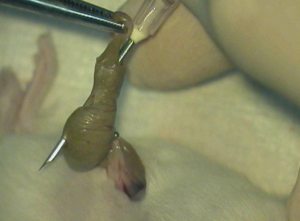
The C.L.P. surgery is performed on Day 0 of the study. To begin, the mice are put under anesthesia, and a small incision is made in the middle of the shaved area. The cecum is located within the peritoneal cavity and is carefully pulled through the incision and fully externalized. Care is taken to minimize the rupturing of blood vessels when the cecum is being moved in or out of the body. Approximately 70% of the cecum is ligated with a clip to restrict blood flow to the area and prevent any more material from entering. The cecum is then punctured with a needle of a predetermined size specified in the protocol based on the desired level of disease state. The larger the needle, the more severe the resulting infection becomes.
By puncturing the cecum, the food material that’s inside will then leak out into the body cavity, creating a septic infection in the mice. After puncturing, the cecum is carefully returned to the peritoneal cavity, which is then sutured back together again. The skin around the incision is stapled together, and then a dab of lidocaine is applied to the surgical site. Each mouse receives a 0.5ml subcutaneous injection of saline and may also have their body weight taken at this time. Finally, the mice are returned to their home cage under a heat source to recover from anesthesia.
Post-Surgery Procedure (Day 0 – End of Study)
After surgery, mice may begin receiving treatments based on the schedule specified in the study protocol. If there are no treatments to be performed, then the mice will simply be observed for a week or so. Clinical signs, body weights, and animal mortality are tracked during this time. If treatments were performed, then comparisons can be made between the different treatment groups based on overall survival rates or differences in relative body weights. If biomarkers are an endpoint for the study, then survival blood draws may be performed on the animals at various times post-treatment. Blood samples can be shipped back to the client or sent to a third party for biomarker analysis.
Why is C.L.P. an Important Model?
Septic infections are very dangerous and potentially deadly in humans and can happen for any number of reasons. Therefore, we are always seeking to discover new ways to prevent and treat these infections. This model is intended to replicate a septic infection that a human might get to test novel antibacterial drugs that may help fight the infection and save lives. Creating the infection in mice provides a close comparison to humans of how this infection progresses in a living organism with an immune system.
From Preclinical Data to Clinical Trials
The overall goal of these studies is to provide a dataset that shows one or more novel treatments being safe to take and effective against the infection in one group, compared to a group of individuals with the infection who receive no treatment. Only after multiple sets of reliable, statistically significant, and independently verifiable data demonstrating the safety and effectiveness of a particular drug have been generated may it have the potential to be tested in human (clinical) trials. If it can pass those trials as well, then your drug may very well be the next lifesaving drug on the market.
If this model is of interest to you or you’d like to learn more, contact TransPharm to discuss your study needs. To hear what others have said about working with us, view our client testimonials.